
C2H2 lewis dot structure
C2H2 Molecular Geometry / Shape and Bond Angles (see description for note) Wayne Breslyn 725K subscribers Join Subscribe Subscribed 395 Share Save 121K views 10 years ago A quick explanation of.

How to Draw the Lewis Dot Structure for C2H2 Acetylene (Ethyne) YouTube
In the C 2 H 2 Lewis structure, there is a triple bond between the two carbon atoms, and each carbon is attached with one hydrogen atom, and none of the atoms has a lone pair. Contents Steps #1 First draw a rough sketch #2 Mark lone pairs on the atoms #3 Calculate and mark formal charges on the atoms, if required
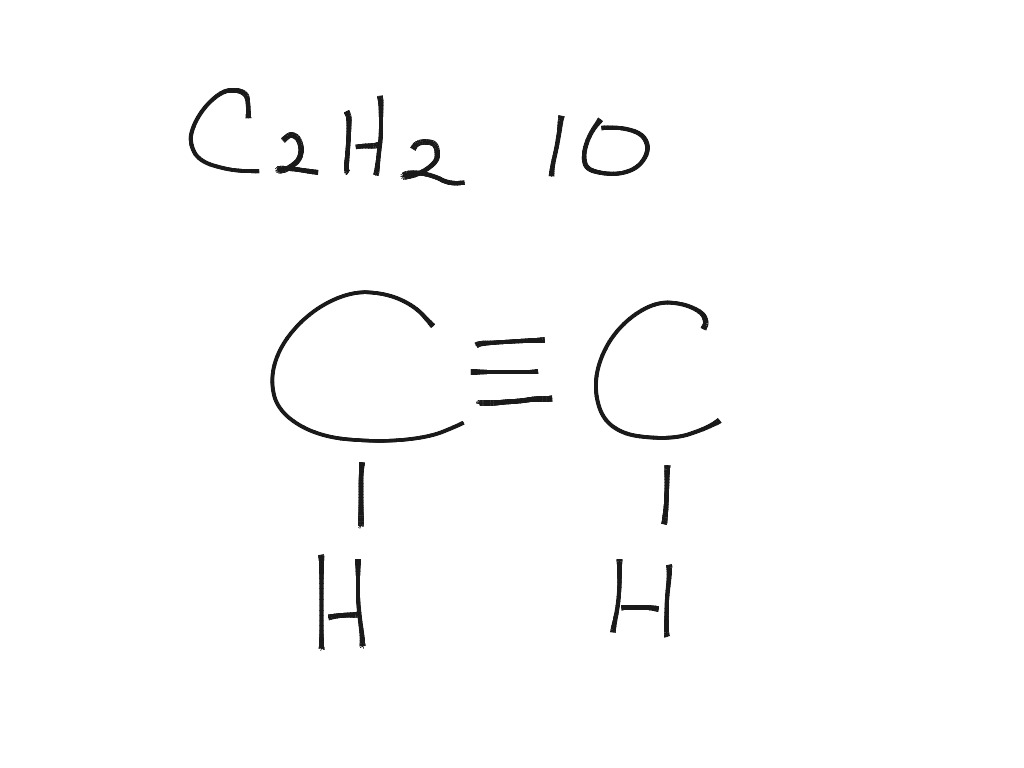
C2h2 Lewis Structure
C2H2 or Ethyne is a nonpolar molecule. It has a symmetric distribution of electrons and a linear geometry. The polarity of any molecule depends on the following factors: C2H2 is made up of two types of atoms: Carbon and Hydrogen. The Carbon atom has an electronegativity of 2.55 and Hydrogen has an electronegativity value of 2.20.

C2H2 Molecular Geometry / Shape and Bond Angles YouTube
C 2 H 2 (acetylene or ethyne) contains two carbon atoms and two hydrogen atoms. There is a triple bond between carbon atoms and hydrogen atoms are joint with carbon atoms though sigma bonds. There are no lone pairs on carbon or hydrogen atoms. In this tutorial, we are going to learn how to draw the lewis structure of C 2 H 2 step by step.

How do you draw the Lewis structure for C2H2? Ethyne or Acetylene
The Lewis structure for C2H2, also known as ethyne or acetylene, is a diagram that shows the arrangement of valence electrons and the bonding between atoms in a molecule. This structure is essential in understanding the properties and behavior of C2H2 in chemical reactions.

Lewis electrondot structure of C2H2 YouTube
A. Definition and concept The C2H2 Lewis structure refers to the arrangement of atoms and electrons in a molecule of ethyne (C2H2) using Lewis dot diagrams. This involves representing each atom using its chemical symbol and drawing dots around it to represent its valence electrons.

C2H2 Lewis Structure (Ethyne or Acetylene) in 2021 Lewis, Molecules
Learn the steps to draw the Lewis Structure of C2H2 (ethyne or acetylene) in just 1 minute.📌You can draw any lewis structures by following the simple steps.

Estrutura De Lewis Do C2H2 Detalhes científicos
Contents show Lewis Structure of Acetylene (C2H2) Lewis Structure is the pictorial representation showing how the valence electrons are participating in bond formation. To study this, first, it is crucial to know the electronic configuration of the participating elements.
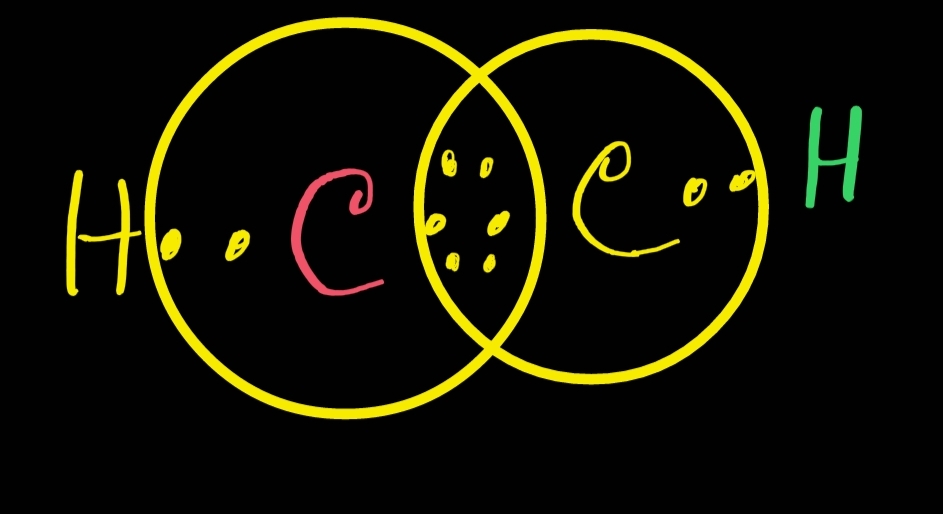
C2H2 Lewis structure ,Valence Electrons, Formal Charge
Step 1: Figure out how many electrons the molecule must have, based on the number of valence electrons in each atom. When drawing the structure of an ion, be sure to add/subtract electrons to account for the charge. Step 2: Connect the atoms to each other with single bonds to form a "skeleton structure.".

Estructura De Lewis De C2h2 Blogan
27K views 1 year ago A step-by-step explanation of how to draw the C2H2 Lewis Dot Structure (Acetylene (Ethyne)). For the C2H2 structure use the periodic table to find the total number of.

C2H2 Lewis Structure, Molecular Geometry, Hybridization, Polarity, and
2.1K 282K views 10 years ago A step-by-step explanation of how to draw the C2H2 Lewis Dot Structure (Ethyne or Acetylene). For the C2H2 structure use the periodic table to find the total number.

C2H2 Lewis structure ,Valence Electrons, Formal Charge
Follow these simple steps to draw Lewis dot structures: Draw the atoms on paper and put dots around them to represent valence electrons of the atom. Be sure to have the correct number of electrons. If the species is an ion, add or subtract electrons corresponding to the charge of the ion. Add an electron for every negative (-) charge, and.
Estrutura De Lewis C2h2 AskSchool
I quickly take you through how to draw the Lewis Structure of CHCH (Acetylene or ethyne). I also go over hybridization, shape, sigma, pi bonding and bond ang.

C2h2 Lewis Structure Vsepr Fotodtp
Lewis structure of C2H2 (or Acetylene or Ethyne) contains one triple bond between the two Carbon (C) atoms and two single bonds between Carbon (C) & Hydrogen (H) atoms. Let's draw and understand this lewis dot structure step by step. (Note: Take a pen and paper with you and try to draw this lewis structure along with me.
C2h2 Estructura De Lewis Blogan
In drawing the Lewis structure for C 2 H 2 (also called ethyne) you'll find that you don't have enough valence electrons available to satisfy the octet for each element (if you use only single bonds). The solution is to share three pairs of valence electrons and form a triple bond between the Carbon atoms in C 2 H 2 .

41 lewis dot diagram for c2h2 Diagram For You
C 2 H 2 is the chemical formula of the simplest and the first member of the alkyne family. It is most commonly known as acetylene in the scientific world. However, the IUPAC name for acetylene is ethyne. It exists as a colorless gas (molar mass = 26.04 g/mol) at room temperature and has a garlic-like odor.